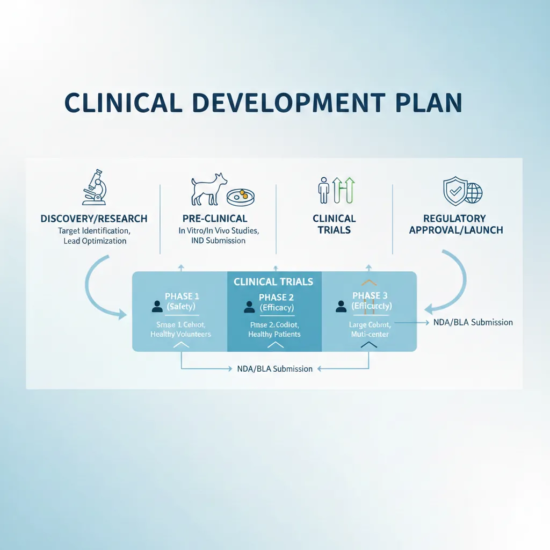
VGCRO assists in developing structured, phase-appropriate clinical development plans.
Scope Includes:
- Development of clinical development roadmaps
- Alignment of individual trials with long-term objectives
- Identification of development risks and mitigation strategies
- Integration of regulatory and operational considerations
Sponsor Value:
✔ Coherent development pathway
✔ Reduced amendments and delays
✔ Clear clinical direction
