
VGCRO provides clinical strategy support to help sponsors define the clinical positioning..
VGajjar Clinical Research Private Limited (VGCRO) is an India based, globally oriented full-service Clinical Research Organization (CRO) providing end-to-end clinical research solutions across the drug and medical product development lifecycle.
Built on a strong foundation of science, ethics, and regulatory compliance, VGCRO supports pharmaceutical, biotechnology, and healthcare organizations in conducting high-quality, inspection-ready clinical trials in line with global regulatory expectations.

VGCRO provides clinical strategy support to help sponsors define the clinical positioning..

We support sponsors in translating non-clinical and early human data..
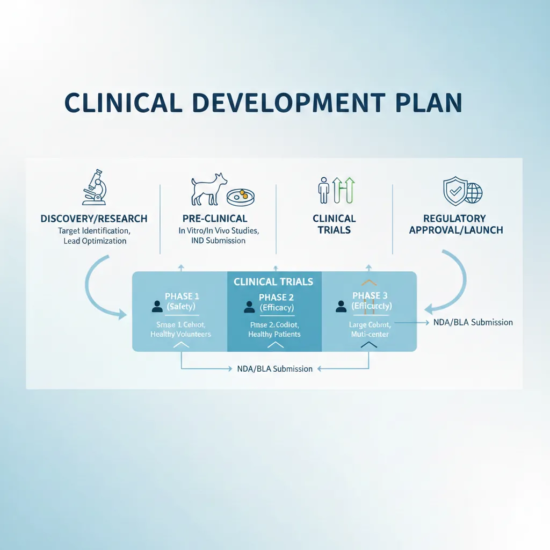
VGCRO assists in developing structured, phase-appropriate clinical..




Our monitoring activities ensure protocol adherence, data quality, and subject protection..

Quality oversight is embedded in all VGCRO activities. While certain technical services are..

Delivered through validated partner platforms and teams..

Statistical services provided by experienced partner statisticians..
Founded
Therapeutic Areas
Cumulative Experience
Develop Standard Operating Procedures (SOPs) and ensure regulatory alignment.

Conduct internal and external audits to identify compliance gaps.

Update processes, SOPs, and provide feedback for quality enhancement.



Develop Standard Operating Procedures (SOPs) and ensure regulatory alignment.

Conduct internal and external audits to identify compliance gaps.

Update processes, SOPs, and provide feedback for quality enhancement.
